Building Trust and Transparency in Healthcare Data: Veritas Supports FAIR Principles and FDA Guidance
At Veritas, we firmly believe in the power of data to drive innovation and improve outcomes in healthcare. But that power will only be realized if that data is trusted by users and regulators. That’s why we are deeply committed to supporting principles and guidelines that foster transparency, traceability, and trust in the data we provide, including the FAIR Principles and the FDA’s emerging transparency guidance.
The evolving regulatory landscape demands a higher level of scrutiny and accountability in how healthcare data is handled. By aligning with FAIR (Findable, Accessible, Interoperable, Reusable) and the FDA’s focus on data quality and transparency, we ensure that our customers can conduct responsible research and navigate regulatory submissions with confidence. We believe that adopting these practices is not just about compliance; it’s about driving better science and ultimately, better patient care.
Here’s how Veritas embodies the FAIR principles:
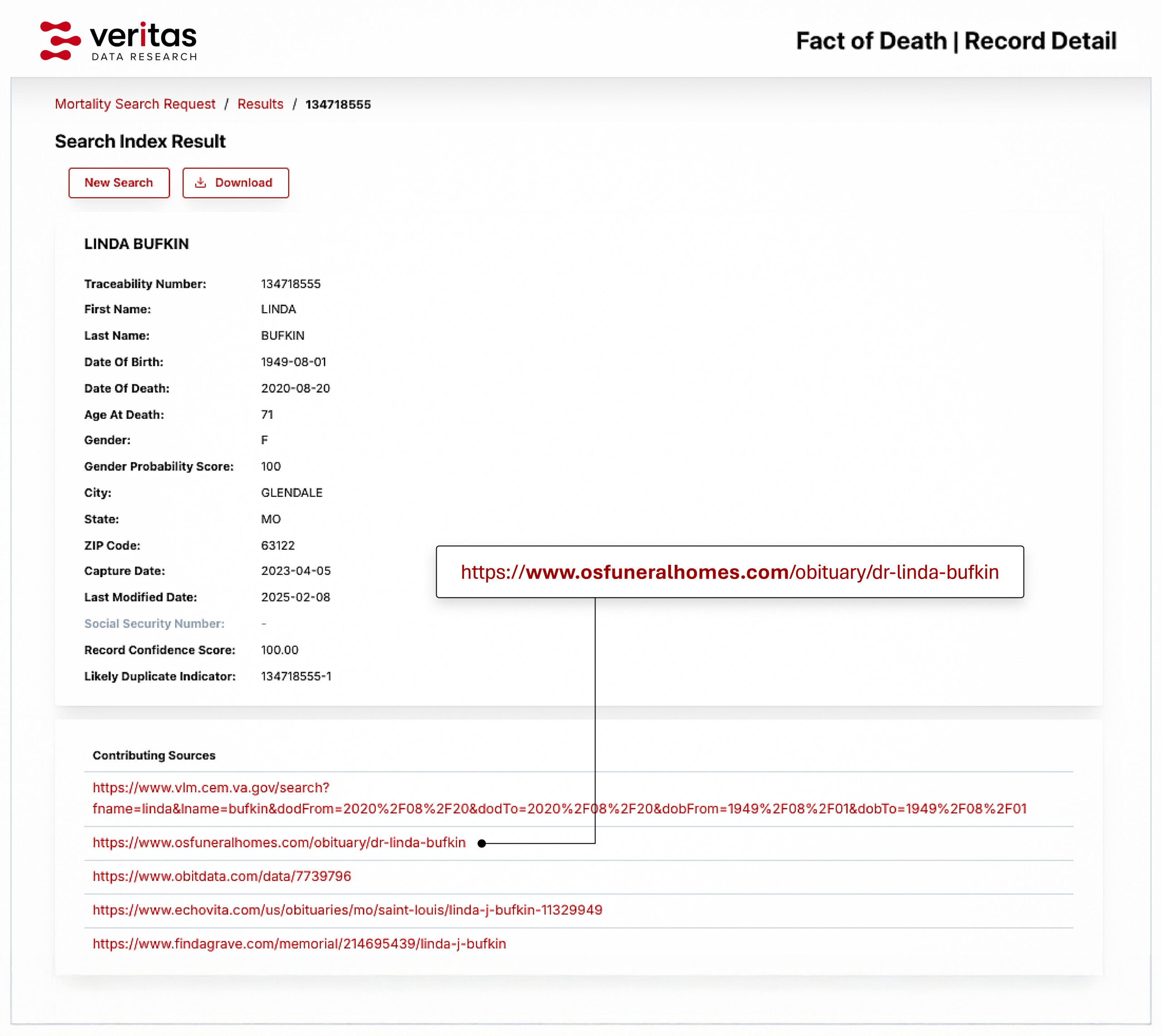
• Findable: Every source record in our dataset is assigned a globally unique traceability number and linked detailed metadata for where that data has been sourced from. Data is ingested through dedicated pipelines for collection, extraction and standardization, ensuring clear categorization and identification. This ensures data and individual data values are consistently organized, easy to discover and traceable throughout the process.
• Accessible: We understand the importance of controlled yet seamless access to data so we provide data and metadata through secure, standard protocols across multiple environments All data is traceable back from delivery to raw sources with unique ID, providing a clear lineage and audit , to the original sources. Access conditions are well defined, ensuring that authorized users are reliably provided the data they are entitled to. We also support data archiving requirements, to allow preserving of reference datasets for publications and regulatory submissions.
• Interoperable: To facilitate seamless integration and analysis, our data uses shared vocabularies, ontologies, and well-documented formats, while also being standardized to common reference datasets. Beyond technical formats, we provide field-level transparency to our methodology: documenting how each attribute is derived, where each record originated, and how standardization and cleaning have been applied. This clarity and transparency ensures that fields are consistently mapped, well understood, and interoperable across research, clinical, and regulatory applications.
• Reusable: We ensure that every dataset comes with comprehensive provenance details, tracing the origin of each record, the sources that contributed, and the transformations applied during processing. This transparency provides researchers with the context and information they need to determine reliability of the data they are using, and where necessary, replicate and validate that data with confidence.
The FDA’s increasing emphasis on data quality, traceability, and transparency in clinical research further reinforces the importance of these principles. Veritas believes that our practices align well with their guidance on data standards and the need for transparent, reproducible, and explainable use of data, and that our mortality data and underlying processes are robust enough to support real-world evidence submissions to FDA.
For more information on how Veritas Data Research can support your data needs and compliance efforts, please contact us at support@veritasdataresearch.com.
Related Articles
View all
Unlocking Opportunity in the Great Wealth Transfer

Datavant and Veritas Expand Partnership to Improve Access to Mortality Data for Healthcare Research

Drug Safety and Real World Data (RWD)-Derived Cause of Death
Request More Information
Speak to a Veritas expert to learn how subscribing to our data can make your organization’s operations and analytics more effective.

